FDA could grant emergency use authorization of Pfizer’s vaccine as early as Friday.
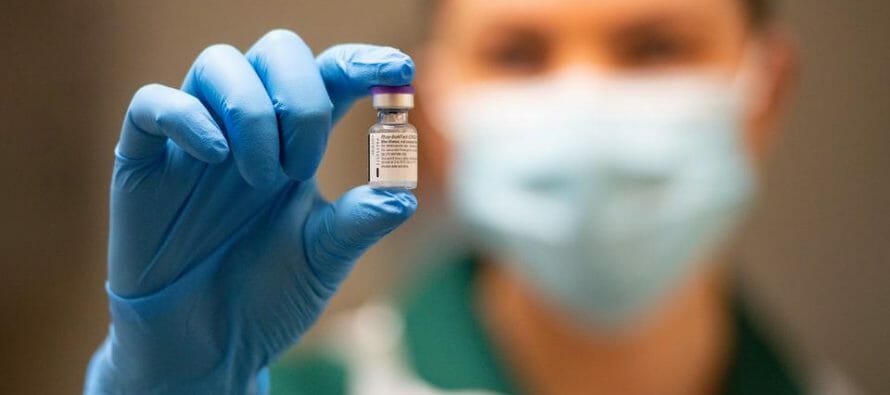
Key committee endorses Pfizer’s COVID-19 vaccine, paving way for clearance by FDA and vaccinations in US to begin in mass.
A committee of leading U.S. vaccine scientists recommended Thursday that the Food and Drug Administration authorize the first COVID-19 vaccine for Americans.
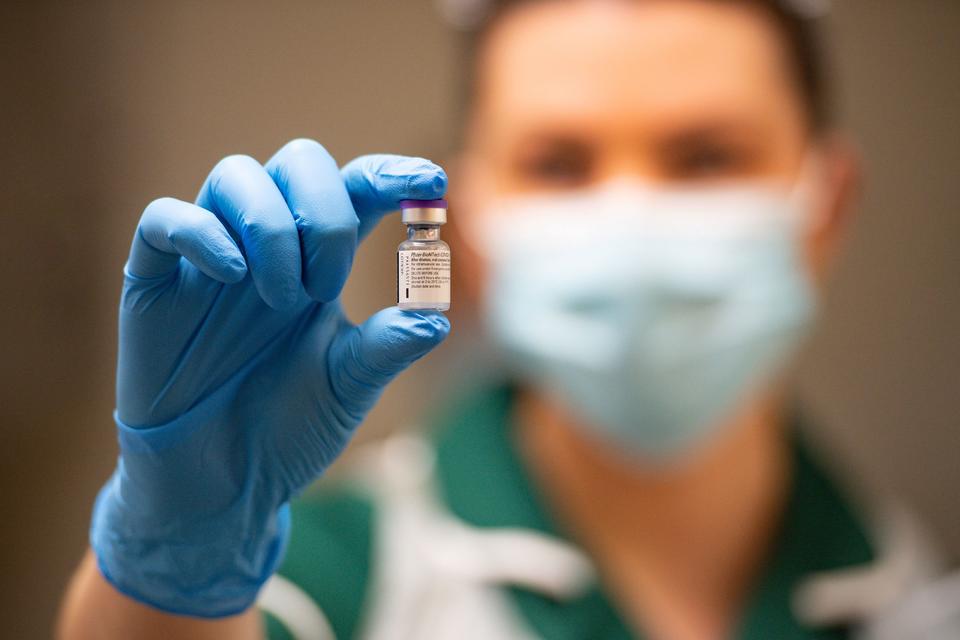
The endorsement paves the way for a final decision by the FDA and mass vaccinations to begin within days in thousands of front-line heath care workers and nursing homes residents.
Members found the benefits of the vaccine outweighed the risks for most people 16 years old and older.
With the vote made, the committee’s recommendation now goes to the FDA, which could authorize the vaccine for emergency use as early as Friday. Once it’s authorized, vials of the vaccine will begin shipping to all 50 states.
Key Points
The full FDA could grant emergency use authorization of Pfizer’s vaccine as early as Friday.
The Vaccines and Related Biological Products Advisory Committee plays a key role in approving flu and other vaccines in the U.S.
The FDA doesn’t have to follow the advisory committee’s recommendation, but the agency often does.
Related Articles
The Living Desert Zoo & Gardens remains open, wildlights, hiking trails accessible, gift shop remains open inside.
Gyms, restaurants, movie theaters and places of worship are among the entities affected by Newsom’s order placing The Coachella
Riverside, San Bernardino and Ventura counties, No New Deaths from Coronavirus Thursday
Riverside, San Bernardino and Ventura counties all reported no new deaths attributed to COVID-19 Thursday. Riverside County health officials reported 83
California Rules On Gatherings: Limit To Three Families, Two Hours Or Less, No Singing
California rules on Large Gatherings to Limit to Three Families, Two Hours Or Less, and No Singing COVID-19, Gov. Gavin