Covid Vaccine: UK Woman First In World To Receive Pfizer Shot, Begins Mass Vaccination Era (Video)
by Publisher CoachellaValley | December 8, 2020 1:06 pm
Covid vaccine: UK woman becomes first in world to receive Pfizer shot Margaret Keenan, 90, given approved vaccine to start mass coronavirus immunization program.
A 90-year-old woman has become the first patient in the world to receive the Pfizer Covid-19 vaccine after its approval in the UK, where the NHS has launched its biggest vaccine campaign.
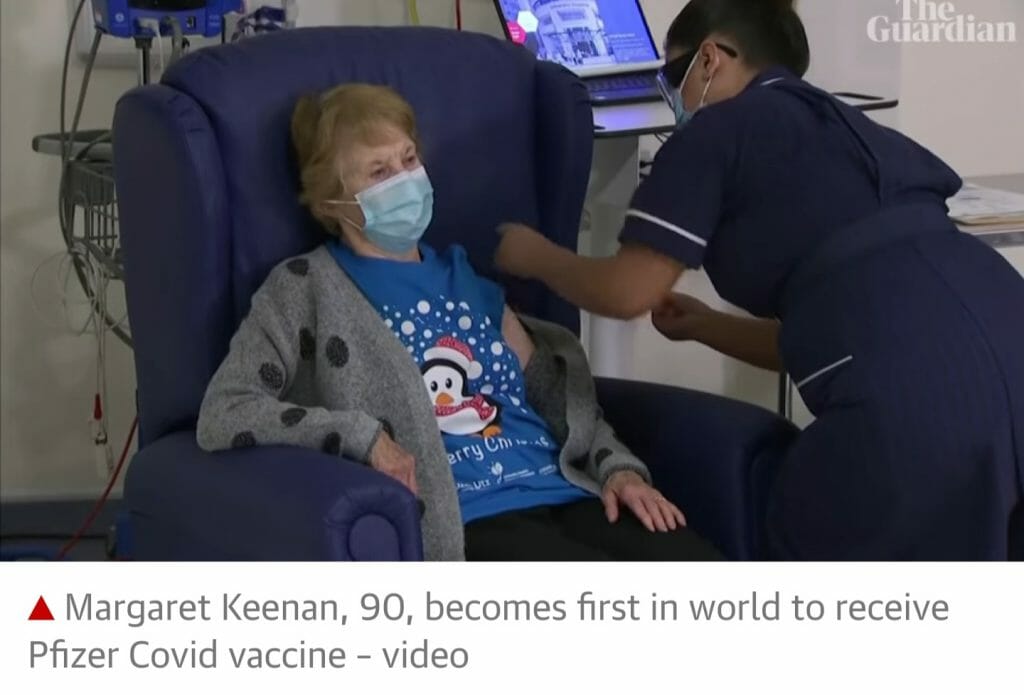
Margaret Keenan received the jab at about 6.45am in Coventry, marking the start of a historic mass vaccination program.
The vaccines will be administered at 50 hospital hubs around the country, with patients aged 80 and over who are either already attending hospital as an outpatient or are being discharged home after a hospital stay, being first in line.

Keenan, known as Maggie, received the injection from the nurse May Parsons at University hospital and said it was a “privilege”.
The former jewellery shop assistant who retired four years ago turns 91 next week.
“I feel so privileged to be the first person vaccinated against Covid-19, it’s the best early birthday present I could wish for because it means I can finally look forward to spending time with my family and friends in the new year after being on my own for most of the year,” said Keenan, who has a daughter, a son and four grandchildren.
U.S. FDA scientists to report Pfizer vaccine safe and effective:
After spending weeks poring over data provided by Pfizer, government scientists on Tuesday acknowledged in a new document that they believe there is ample evidence that the first COVID-19 vaccine works and is safe in people over the age of 16.
While that assessment isn’t the coveted green light the nation has been waiting for, the briefing suggests agency staff aren’t warning of any last-minute hiccups and that an emergency authorization is well within the nation’s grasp.
It also suggests that the vaccine might not just be limited to people over the age of 18 as was initially expected. And the government found protection was strong after just one dose, though both doses are recommended for the strongest protection.MORE: What to know about COVID-19 [3]vaccines [4]and how they work
“Safety data from approximately 38,000 participants [greater than or equal to] 16 years of age randomized 1:1 to vaccine or placebo with a median of 2 months of follow up after the second dose suggest a favorable safety profile, with no specific safety concerns identified that would preclude issuance of an EUA,” the FDA wrote.
The report notes the most common reaction was a skin reaction at the injection site, followed by fatigue or headaches. About half of recipients between 16 and 55 experienced side effects like fatigue or headaches and about a third reported chills or muscle pain. Of clinical trial participants over 55 about half reported fatigue, a third reported headache, and about a quarter reported chills or muscle pain.
The data also indicates the vaccine works equally men in men and woman, and among people of different races and ethnicities.
FDA noted there are some areas where there isn’t enough data on the Pfizer vaccine, such as how it works in people with medical conditions like HIV/AIDS, in people who have recovered from COVID-19 infection, or how long the protection lasts.
The report also highlights that while the Pfizer vaccine prevents recipients from getting sick with symptoms of COVID-19, there isn’t enough data yet to know if the vaccine is effective enough at preventing all transmission of the disease that would eventually replace the need to wear masks, socially distance, and avoid large, indoor gatherings.
So even if you are an early recipient of the vaccine it will be important to maintain these risk lowering techniques to protect others.
- Protesters Descend on El Paseo Drive, Restaurants Stay Open In Defiance of New Order: https://coachellavalley.com/protesters-descend-on-el-paseo-drive-restaurants-stay-open-in-defiance-of-new-order/
- Can Gyms Stay Open Under California’s New Stay-At-Home Order?: https://coachellavalley.com/can-gyms-stay-open-under-californias-new-stay-at-home-order/
- MORE: What to know about COVID-19 : https://abcnews.go.com/Health/covid-19-vaccines-work/story?id=73796121
- vaccines : https://abcnews.go.com/alerts/vaccinations
Source URL: https://coachellavalley.com/covid-vaccine-uk-woman-first-in-world-to-receive-pfizer-shot-begins-mass-vaccination-era-video/